Customer Updates: Stryker Network Disruption > Read Now
Spine
We deliver the spine technologies, empowering our customers to keep the world moving. Together, we do more to challenge the standard, respond to clinical needs and pioneer new possibilities.

Balance ACS (BACS)
Balance ACS is a comprehensive platform that applies three-dimensional solutions across the entire clinical care continuum to help drive quality outcomes for spine patients. It provides tools – from surgical preauthorization and preoperative planning to 3D anatomical modeling and postoperative reporting – to support the perioperative process.
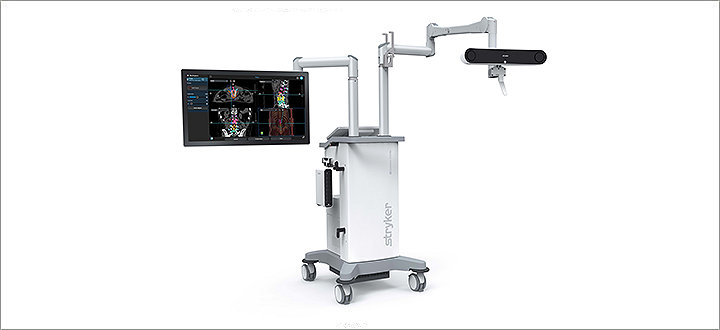
Spinal and cranial navigation
Featuring optical tracking options, sophisticated software algorithms and instrumentation specifically designed for spine surgery, today’s Q Guidance System aims to deliver more surgical spine and cranial planning and navigation capability than ever before.
We are working hard to make healthcare better by partnering with our customers to help develop, implement and sustain patient-centric spine service lines.
We can help bring you closer to your clinical, operational and financial goals
Looking for a product IFU?
NS-GSNPS-SYK-3301297_REV-0
