Customer Updates: Stryker Network Disruption > Read Now
Biologics
Stryker’s advantage
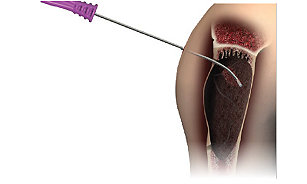
Stryker is one of the world’s leading medical technology companies. In order to better partner with our customers, we made the decision to invest in the development of our biologics portfolio to bring a comprehensive solution to a variety of specialties, including orthopaedic, spine, sports medicine, and CMF.
Stryker continues to develop innovative technologies to enhance our spectrum of products, while collaborating with industry-leading tissue organizations to offer a robust portfolio of proprietary spinal grafts, viable bone matrices, demineralized bone matrix products, synthetic bone grafting substitutes, amniotic and dermal membranes, and nerve products.
Our focus on actively bringing new and compelling products to our portfolio is guided by our commitment to both surgeon and patient needs, as well as a quality-first foundation. Stryker works with various heath authorities and regulatory bodies, such as the U.S. Food and Drug Administration (FDA) and organizations like the American Association of Tissue Banks (AATB) to ensure our products meet the appropriate standards.
CP-AWI-47