Customer Updates: Stryker Network Disruption > Read Now
Reinforced anesthesia needle
ENT surgeons select the reinforced anesthesia needle for its innovative design.1-2
The unique combination of a 27G tip and larger outer sleeve allows for a positive stop for injection depth precision3-4

Features and benefits
The reinforced anesthesia needle was designed to offer ENT surgeons enhanced control. Important features and benefits include:
- The unique combination of a 27G tip and larger outer sleeve allows for a positive stop for injection depth precision1,4
| Product | Ordering no. | Description |
| Reinforced anesthesia needle | RAN-027-2 | 27 G, 3.5", 2-pack (only sold in a bundle configuration) |
| Reinforced anesthesia needle | RAN-027-5 | 27 G, 3.5", 5-pack |
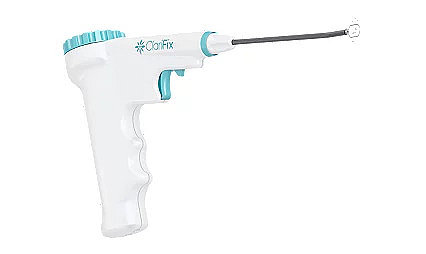
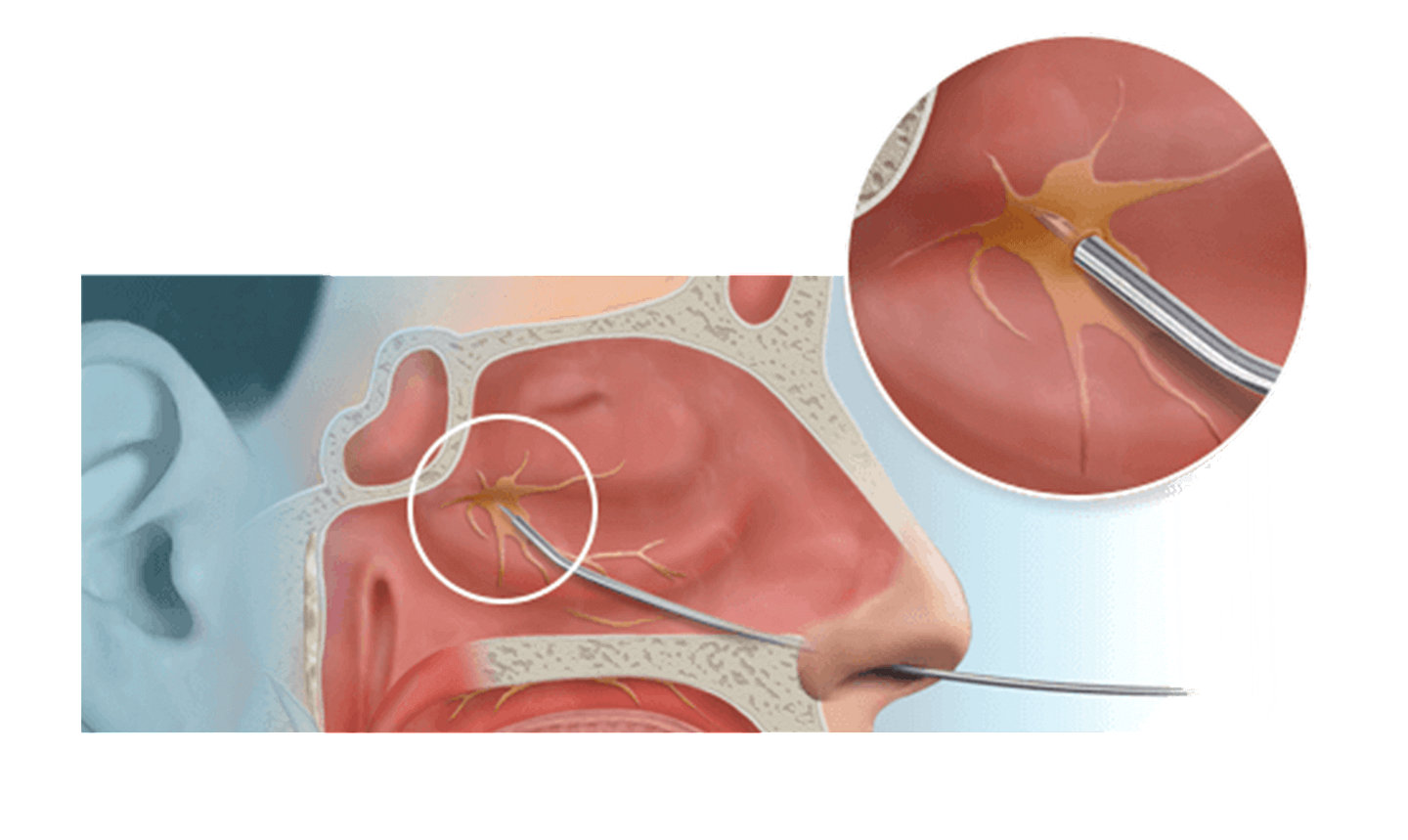
ClariFix® cryotherapy device
Stryker's ClariFix device goes straight to the root of chronic rhinitis - the overactive nerves. Using a minimally invasive treatment called cryotherapy, the ClariFix device applies focal, controlled freezing to target tissue under direct visualization.7

XprESS® ENT dilation system
This system delivers control and versatility for balloon dilation. Its seeker-based design and tip malleability provide the tactile feel and flexibility to address complex anatomy for a broad range of patients.5-6
References:
- Data on file at Stryker 3455-004
- Data on file at Stryker 3455-006
- Data on file at Stryker 4674-001
- Reinforced anesthesia needle instructions for use 5117-001
- XprESS instructions for use 4322-001
- Data on file at Stryker DHF 1656-001
- ClariFix instructions for use 5082-001
This document is intended solely for the use of healthcare professionals. A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. We do not dispense medical advice and recommend that surgeons be trained in the use of any particular product before using it in surgery.
ENT-INST-SYK-619096


