Customer Updates: Stryker Network Disruption > Read Now
Stryker Launches New Tendon Fixation Device System
15-Sep-2021
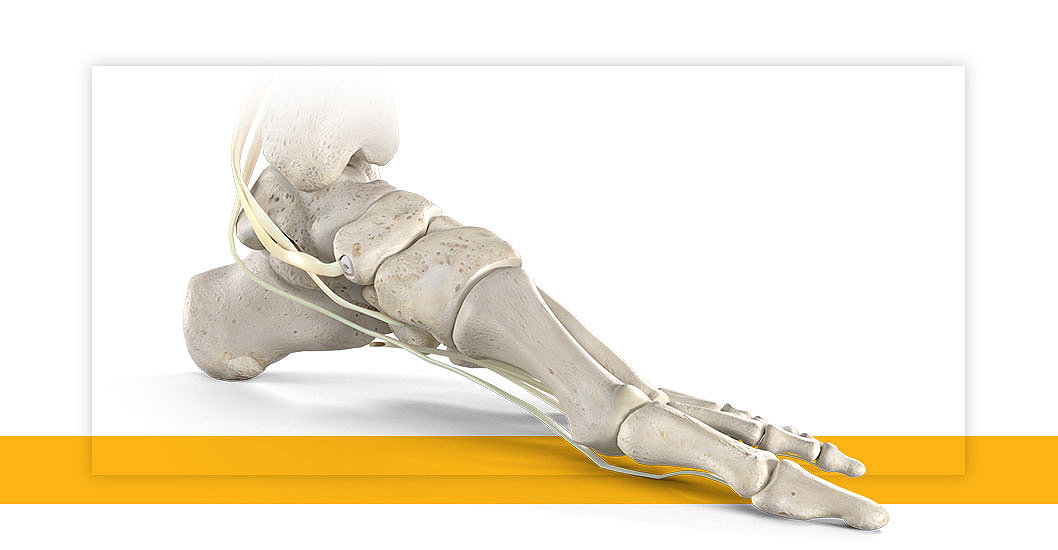
MAHWAH, N.J.—Stryker’s Trauma & Extremities division has launched its Citrelock™ Tendon Fixation Device System. The new system provides surgeons a differentiated design via a tendon thread featuring a resorbable technology, known as Citregen™, that has unique chemical and mechanical properties for orthopaedic surgical applications. Stryker will debut Citrelock at the American Orthopaedic Foot & Ankle Society (AOFAS) annual meeting, Sept. 22-25, 2021, in Charlotte, N.C.
“Our customers are looking for a more predictable and effective bioresorbable material than what is currently on the market,” said Michael Rankin, vice president, marketing and medical education of Stryker’s Foot & Ankle business. “The Citrelock Tendon Fixation Device System helps fill that need with its innovative material technology and unique design. Citregen is an exciting addition to Stryker’s existing biomaterial portfolio and will be expanded for use in additional Trauma & Extremities indications in the future.”
The Citrelock Tendon Fixation Device System offers surgeons:
- A controlled and homogeneous resorption process that prevents bulk degradation and chronic inflammation.1
- Compressive strength that is comparable to cortical bone with a modulus comparable to cancellous bone.1,2
- Citregen contains citrate, calcium, and phosphate molecules that are inherent to the bone anatomy.1
- Material polymer structure that mimics the extracellular matrix protein network.1
- Citregen maintains structural integrity during the healing phase, while the implant is replaced by host tissue over time .1
"By leveraging Citregen’s unique material properties, the Citrelock Tendon Fixation Device System introduces design features to ease insertion and secure fixation without damaging the tendon during placement,” said Wayne Berberian, MD, orthopedic surgeon at HMH Hackensack University Medical Center. “It has been exciting to observe the development of a biomaterial for use in orthopaedic applications. I’m excited to start using Citrelock in my practice.”
Citrelock will be featured during one of Stryker’s industry sessions at AOFAS on Sept. 24. The company is also hosting a new product reception highlighting the benefits of Citrelock along with other recent additions to its Foot & Ankle portfolio. For more information, visit www.stryker-trauma.live/aofas-2021/ or stop by the Stryker booth (No. 425).
About Stryker
Stryker is one of the world’s leading medical technology companies and, together with its customers, is driven to make healthcare better. The company offers innovative products and services in Orthopaedics, Medical and Surgical and Neurotechnology and Spine that help improve patient and hospital outcomes. More information is available at www.stryker.com.
Follow Stryker Foot & Ankle on LinkedIn and Stryker’s Trauma & Extremities division on Twitter.
Media Contacts
Andrea Sampson, Sampson Public Relations Group
asampson@sampsonprgroup.com
562.304.0301
References:
1. FDA 510(k) K200725
2. TM-210202
A surgeon must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. Stryker does not dispense medical advice and recommends that surgeons be trained in the use of any particular product before using it in surgery.
The information presented is intended to demonstrate the breadth of Stryker’s product offerings. A surgeon must always refer to the package insert, product label and/or instructions for use before using any of Stryker’s products. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your sales representative if you have questions about the availability of products in your area.
CITE-BL-1, 08-2021
