Customer Updates: Stryker Network Disruption > Read Now
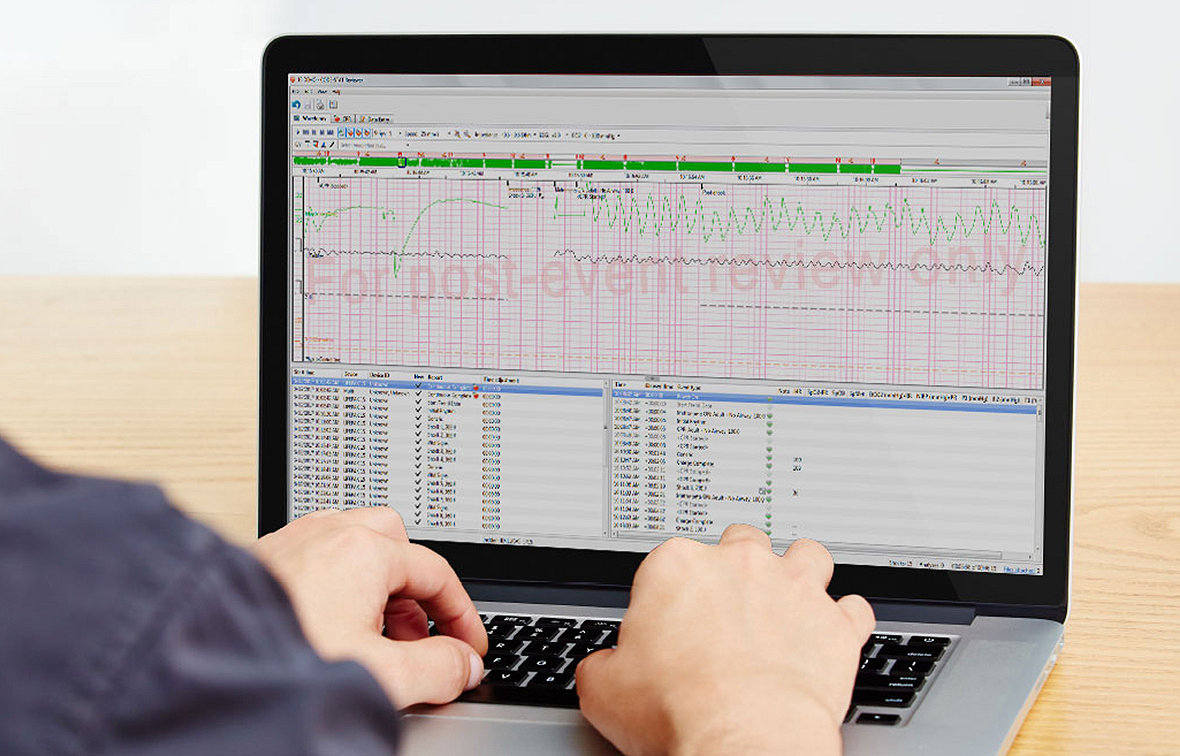
CODE-STAT data review software
CODE-STAT data review software
Insights to improve performance and patient outcomes
Turn your passion for saving lives into targeted improvements — CODE-STAT data review software and service lets you easily understand team performance immediately after response.
Product highlights
Better data means better CPR
CODE-STAT software makes it easy to see and correlate key CPR performance metrics while the incident is still fresh. Spot at a glance where you need to improve — or gain confidence in your team’s operations and skills. Measure, review, report, and repeat.

Deliver better care
Respond more effectively for better outcomes with easier access to complete event data.
Train more efficiently
Focus training and performance management where it’s needed most.
Make improvement faster and easier
Simplify the collection, analysis, and sharing of data that can power performance reviews and enhancement.
Save time on data collection and reporting
Collect information automatically and customise required reports and documentation.

Product features
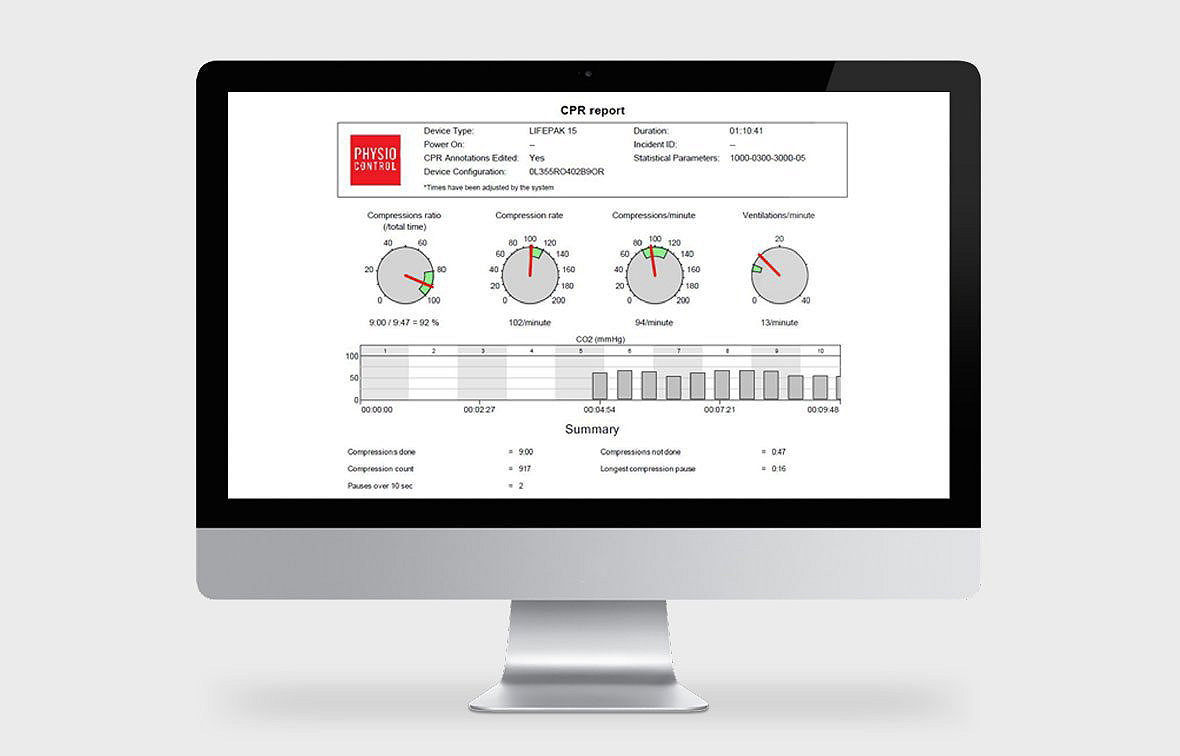
CODE-STATAccelerate your QA/QI efforts with insights revealed through comprehensive CODE-STAT analysis. 2015 ERC guidelines recommendations on CPR quality suggest maintaining “data-driven performance-focused debriefing to improve performance of resuscitation teams.”* CODE-STAT software gives you quick access to relevant high-quality data—including compression hands-on time, chest compression and ventilation, rate, longest pause, peri-shock pause time, and time elapsed before first shock.
*Greif R, Lockey AS, Conaghan P, et al. European Resuscitation Council Guidelines for Resuscitation 2015. Section 10. Education and implementation of resuscitation. Resuscitation. 2015;95:289.
See resuscitation performance at a glance
- Display CPR metrics visually, according to your targets, for intuitive insight. Dive into moment-by-moment details of an entire event on a graphic, continuous time scale.
- Review interval statistics for the entire resuscitation, broken down by time interval.

Create custom analytics and reporting according to your needs
- Customise display of the statistics and details you need most, including CPR performance, ECG and EtCO2 waveforms, and CO2 trend data.
- Annotate the stories of each resuscitation event to add context or focus attention on achievements or areas needing improvement.
- Create individual or summary reports.

Streamline reporting
- Easily create summary, detail, or custom reports for performance reviews, training plans, or documentation.
- Provide feedback to caregivers in a simple, intuitive format.
- Track performance improvement and trends over time.
Reduce data handling effort
- Simplify data collection and management, thanks to full integration with all LIFEPAK monitor/defibrillators and the LUCAS 3 v3.1 chest compression system.

Improved to 83% compression ratio
Speed clinically-significant improvements
Hands-on compression ratio relates directly to patient outcomes, which is why Redmond Medic One set out to increase this performance metric using CODE-STAT software. In just six months, the team increased its compression ratio from 66% to 83% improvement. Research shows clinically significant improvement takes place after the kind of targeted performance reviews you can hold when you learn from every resuscitation with CODE-STAT.
Olasveengen T, Wik L, Kramer-Johansen J, et al. Is CPR quality improving? A retrospective study of out-of-hospital cardiac arrest. Resuscitation. 2007;75:260–266
Take the next step
Want to talk to your Sales Representative? Please complete the form below and we’ll connect you with your local representative.
When you invest in our lifesaving devices and data products, you are buying more than quality design and manufacturing—you’re also buying our unrivaled commitment to customer service and support.
Technical support
We offer one of the largest networks of technical service representatives in the industry. We’ll work with you to quickly assess any issues and find the best solution.
Training and education
As your lifesaving partner, Stryker offers training support, seminars, podcasts, and access to clinical research on the latest resuscitation strategies and tips.
Related products
LIFEPAK 15 monitor/defibrillator
Rely on the LIFEPAK 15 monitor/defibrillator for the confidence you need in emergencies and the highest available escalating energy, up to 360 joules (360J).
Learn moreLUCAS 3, v3.1 chest compression system
Deliver guidelines-consistent, high-quality chest compressions with less strain, micromanagement, and risk for the caregiver. The LUCAS chest compression system provides benefits both to the cardiac arrest patient and the resuscitation team.
Learn moreLIFENET System
The LIFENET System is a comprehensive cloud-based platform that seamlessly manages and delivers patient information and device data that EMS and hospital teams need to work together effectively.
Learn moreThis document is intended solely for the use of healthcare professionals. A healthcare professional must always rely on his or her own professional clinical judgment when deciding whether to use a particular product when treating a particular patient. Stryker does not dispense medical advice and recommends that healthcare professionals be trained in the use of any particular product before using it.
The information presented is intended to demonstrate Stryker’s product offerings. A healthcare professional must always refer to operating instructions for complete directions for use indications, contraindications, warnings, cautions, and potential adverse events, before using any of Stryker’s products. Products may not be available in all markets because product availability is subject to the regulatory and/or medical practices in individual markets. Please contact your representative if you have questions about the availability of Stryker’s products in your area. Specifications subject to change without notice. The products depicted are CE marked in accordance with applicable EU Regulations and Directives.
Stryker Corporation or its divisions or other corporate affiliated entities own, use or have applied for the following trademarks or service marks: LIFEPAK, LIFENET, LUCAS, HealthEMS, RevNet, CODE-STAT, Physio-Control, HeartSine, samaritan, LIFEPAK CR, TrueCPR, REDI-CHARGE, QUIK-COMBO, EDGE, cprINSIGHT, ClearVoice, QUIK-STEP, LIFEPAK TOUGH, HomeSolutions.net, RELI, REDI-PAK, Shock Advisory System, LIFELINKcentral. All other trademarks are trademarks of their respective owners or holder. The yellow and black color scheme is a registered trademark of Stryker Corporation.
LIFEPAK devices: CE Class IIb (0123)
HeartSine device: CE Class IIb (0123)
LUCAS device: CE Class IIb (2460)
EC REP: Stryker European Operations Limited | Anngrove, IDA Business & Technology Park | Carrigtwohill, Co. Cork, T45 HX08 | Ireland
M0000007484 REV AA