LIFEPAK 1000 defibrillator
LIFEPAK 1000 defibrillator
Simple. Flexible. Powerful.
Not every cardiac emergency is the same. Neither is every responder. Your world demands flexibility—and that’s exactly what the LIFEPAK 1000 defibrillator from Stryker delivers.

Simple. Flexible. Powerful.
If you’re first on the scene of a cardiac emergency, the LIFEPAK 1000 AED can help improve lifesaving outcomes and speed the transition of patients to the next critical level of care.
The LIFEPAK 1000 combines the simplicity of an AED with clear guidance, including onscreen and audio prompts with the flexibility of manual mode operation when indicated. Flexible setup options allow on-screen graphics for the basic rescuer or ECG display and access to manual mode for the more advanced rescuer. This powerful AED combines basic guidance with advanced operational control, enabling responders to deliver fast, appropriate treatment.

Product highlights
LIFEPAK 1000In a police and fire rescue vehicle, or out in the field, you’re always ready to respond to cardiac emergencies. The LIFEPAK 1000 provides powerful defibrillation, long battery life, and durability that can stand up to severe environments.

Durable, vehicle-friendly construction
- Endures rigorous drop-tests and includes protective case and bumpers for durability in the harshest environments.
- Designed to ride along in vehicle without damage from continuous vibrations and other movement.

Powerful and easy to use
- Loud voice prompts and on-screen graphics provide clear guidance on applying electrodes and initiating a shock to ensure user confidence.
- Escalating energy to 360 J, biphasic, for difficult to defibrillate patients.
- Provides battery power for up to 220 shocks – escalating to 360 Joules – delivering 17 hours of monitoring time.
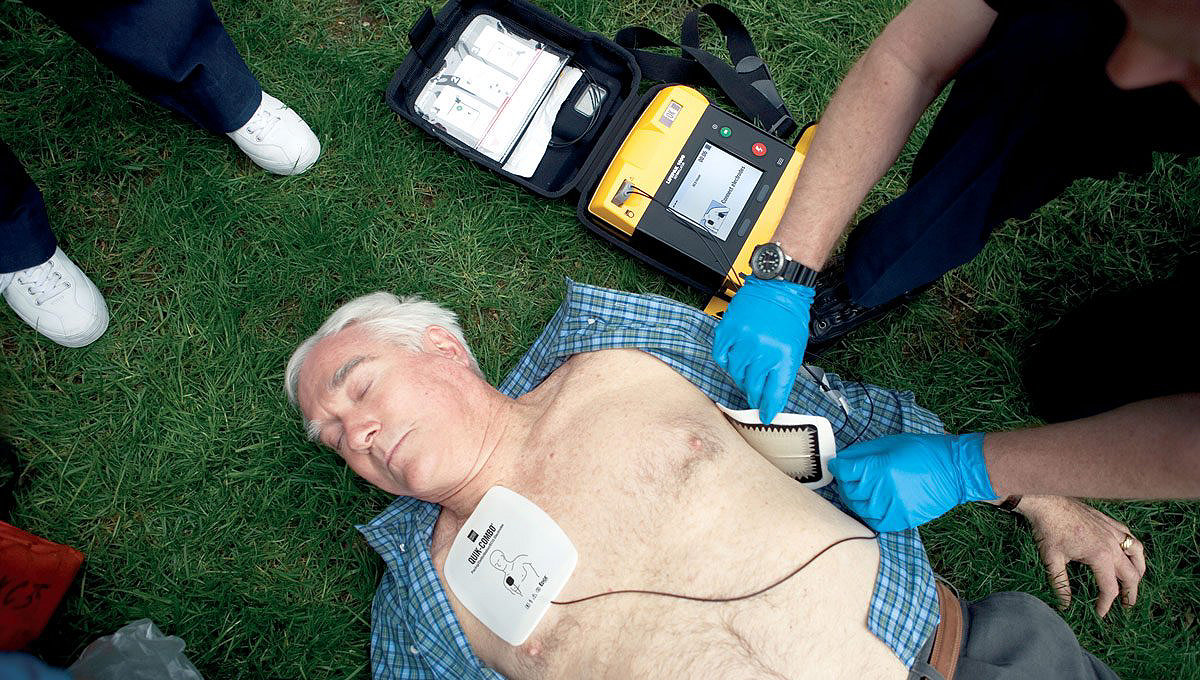
Clinically advanced, with robust features
- Exclusive cprMAX™ Technology minimizes CPR interruptions by allowing responders to perform pre-shock CPR while the AED charges.
- Features including a CPR countdown timer, ECG capability, a shock counter, and programmable settings – all on a large, easy-to-read LCD screen.
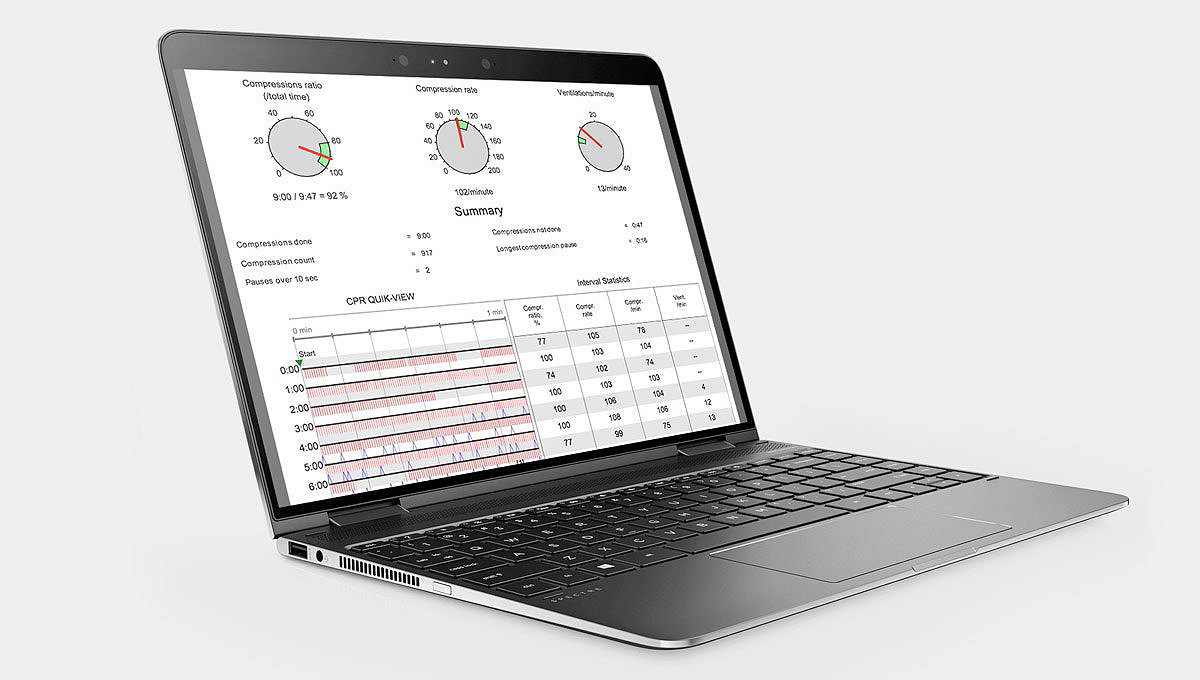
Compatibility and connectivity
Compatibility with other LIFEPAK electrodes and monitors speeds the transition to the next level of care. And, post-event patient and performance data can be easily viewed, analyzed and managed with CODE-STAT data review software – helping to improve future response and target training needs.
Take the next step
Want to talk to your Sales Representative? Please complete the form below and we’ll connect you with your local representative.
When you invest in our lifesaving devices and data products, you are buying more than quality design and manufacturing—you’re also buying our unrivaled commitment to customer service and support.
ProCare services
Our ProCare service plans help ensure the integrity of your Stryker lifesaving system.
Technical support
We offer one of the largest networks of technical service representatives in the industry. We’ll work with you to quickly assess any issues and find the best solution.
Training and education
As your lifesaving partner, Stryker offers training support, seminars, podcasts, and access to clinical research on the latest resuscitation strategies and tips.
Related products
HeartSine samaritan PAD 350P/360P
The compact and easy-to-use HeartSine AEDs empower first responders with lifesaving technology, while integrated Wi-Fi connectivity ensures the devices are ready when needed most.
Learn moreM0000013168 REV AA






