Search Results
Showing results for: clinical study
Showing 50 Of 191 records
-
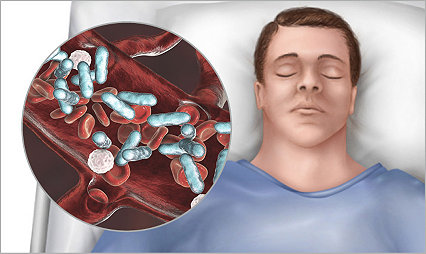
Prevent hospital-acquired pneumonia by reducing any potential risk factors
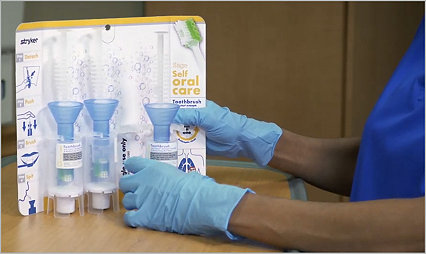
The importance of patient oral care
Hospital oral care plays a pivotal role in reducing the risk of HAP by minimizing the colonization of harmful bacteria in the respiratory tract.
-
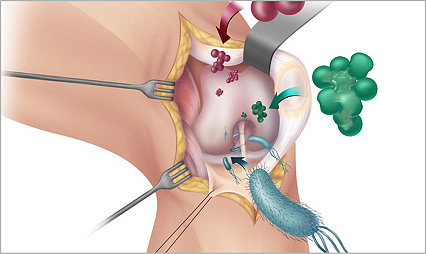
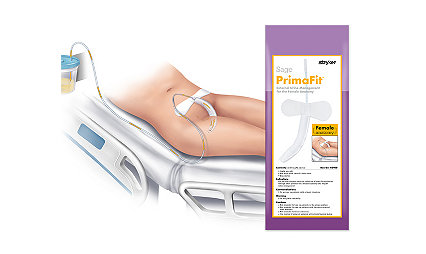
Sage PrimaFit External Urine Management System for the Female Anatomy helped reduce CAUTI rate 63%
Catheter-related infections decreased from before the introduction to EUDFA and after. This is not a cause-and-effect relationship, but the trend is promising.
-
Product resources
Find a variety of documents including best practices, datasheets and clinical information for easy download.
-
Sage 2% Chlorhexidine Gluconate (CHG) Cloths
Clinically proven to address bacteria on the skin
Bacteria on the skin puts your patients at risk for SSIs.
-
Sage Nose to Toes Pre-Op Prepping Systems
Pre-Op Prepping Systems
Clinically proven to address bacteria in the nares, the oral cavity, and on the skin.
-
Sage News and Updates
Sign up to receive product updates, clinically relevant articles and learn about Sage's impact on the healthcare industry and our community.
-
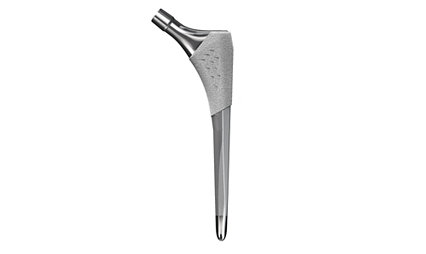
Secur-Fit Advanced
Femoral Hip Stem
Our flagship “fit and fill” femoral stem based on a clinically successful design with modern SOMA-designed refinements*
-
Sage Newsroom
The Sage Newsroom is your source for industry news, clinical information, Sage product and business updates, inspiring stories, and more!
-
Reprocessing and remanufacturing in healthcare
Reprocessing of single-use medical devices (SUDs) is the practice of inspecting, cleaning, function testing, sterilizing and packaging so that they can be clinically and safely used again. Reprocessing of SUDs is regulated by the FDA. We satisfy all FDA regulatory requirements to reprocess medical devices and ensure that our reprocessed devices are substantially equivalent to original equipment devices.
-
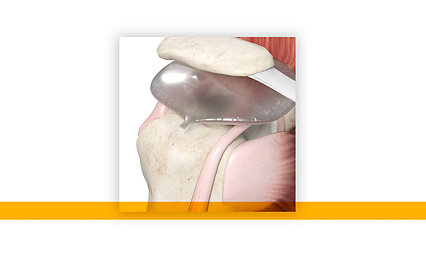
Mako Partial Knee
Mako Partial Knee enables surgeons to create a 3D CT-based surgery plan of their patient's unique anatomy.
-
Dekompressor disc removal system
Our Dekompressor system helps relieve pain caused by contained disc herniations through a procedure called percutaneous discectomy.(1) You can measurably and selectively extract disc material without annular or nuclear disruption.(2,5)
-
Acetabular System
Acetabular System
Have confidence in a powerful combination–a system that builds on the legacy that has defined our Trident brand for more than two decades, paired with the latest additive manufactured Tritanium In-Growth Technology or PureFix HA.
Trident II is created for case-to-case confidence that comes from building on a system backed by years of successful clinical data1-5 and reliable features such as the notable Trident Innerchange Locking Mechanism, Modular Dual Mobility (MDM), and our X3 precisely engineered polyethylene. -
InSpace subacromial balloon spacer
Subacromial balloon spacer
InSpace is the industry’s only minimally invasive biodegradable, subacromial balloon spacer for arthroscopic treatment of massive, irreparable rotator cuff tears (MIRCTs)†.
-
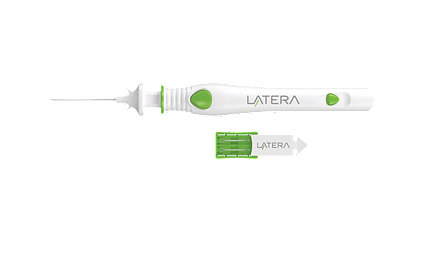
LATERA® absorbable nasal implant system
LATERA supports upper and lower lateral nasal cartilage, which may reduce nasal airway obstruction (NAO) symptoms and help increase nasal airflow.1
-
Sage Comfort Shield Barrier Cream Cloths
Our basinless incontinence care treats and prevents incontinence-associated dermatitis (IAD).
-
ClariFix® cryotherapy device
Over 87 million Americans suffer from chronic rhinitis.1-2 Historically, patients with chronic rhinitis (having symptoms such as frequent runny nose, congestion and or post-nasal drip) have had limited surgical treatment options such as a vidian neurectomy.3
Stryker's ClariFix device goes straight to the root of chronic rhinitis - the overactive nerves. Using a minimally invasive treatment called cryotherapy, the ClariFix device applies focal, controlled freezing to target tissue under direct visualization.4 -
Restoration Modular
Make the most of your revision cases with a clinically-backed platform that is designed to leverage versatility and simplicity to help you achieve desired revision THA outcomes. Keep up with your active patients and rest easy with Restoration Modular. Launched in 2003, there have been over 181,000 Restoration Modular constructs implanted globally; providing solutions for femoral Type 1 through Type 4 revisions.1 This comprehensive system provides the flexibility to help address a broad range of revision scenarios with instrumentation that is designed to facilitate OR efficiency.
-
XprESS® ENT dilation system
The XprESS ENT dilation system delivers a unique combination of control and versatility for balloon dilation. Its familiar seeker-based design and tip malleability provide the tactile feel you are accustomed to and the flexibility you need to address both chronic rhinosinusitis or Eustachian tube dysfunction patients.1-2
-
NeuroMatrix
Connector
Nerve conduits and wraps provide an encasement for peripheral nerve injuries and protection of the neural environment. NeuroMatrix, Neuroflex, and NeuroMend are semi-permeable structures that allow diffusion of nutrients and neurotrophic factors into the conduit, but provide a barrier to larger, scar-forming cells.1,2
-
NeuroMend
Protector
Nerve conduits and wraps provide an encasement for peripheral nerve injuries and protection of the neural environment. NeuroMatrix, Neuroflex, and NeuroMend are semi-permeable structures that allow diffusion of nutrients and neurotrophic factors into the conduit, but provide a barrier to larger, scar-forming cells.1,2
-
Neuroflex
Connector
Nerve conduits and wraps provide an encasement for peripheral nerve injuries and protection of the neural environment. NeuroMatrix, Neuroflex, and NeuroMend are semi-permeable structures that allow diffusion of nutrients and neurotrophic factors into the conduit, but provide a barrier to larger, scar-forming cells.1,2
-
Nerve repair
Peripheral nerve portfolio
Our nerve portfolio consists of type I collagen conduits and wraps. They are designed to provide a protective layer around injured peripheral nerves and offer a resorbable and semi-permeable encasement to facilitate nerve repair.
-
Triathlon
Total Knee System
The Triathlon Total Knee System is a primary total knee replacement system designed to work with the body. Triathlon has been implanted in over 3 million patients worldwide. Triathlon Total Knee is available for use with the Mako System for Mako Total Knee.