Search Results
Showing results for: Additive Manufacturing
Showing 50 Of 201 records
-
Connected OR operating system
OR integration platform
Our OR integration, with its intuitive workflow and safety features, allows nurses to focus on delivering the highest level of patient care rather than managing the nuances of the surgical space.
-
Tritanium C
Anterior Cervical Cage
A hollow implant that consists of a unique configuration of both solid and porous structures built using AMagine Technology, our proprietary approach to implant creation using additive manufacturing
-
Tritanium TL
Curved Posterior Lumbar Cage
A hollow implant that consists of a unique configuration of both solid and porous structures built using AMagine Technology, our proprietary approach to implant creation using additive manufacturing
-
Tritanium PL
Posterior Lumbar Cage
A hollow, rectangular implant that consists of a unique configuration of both solid and porous structures built using AMagine Technology, our proprietary approach to implant creation using additive manufacturing
-
Viju Menon
Group President, Global Quality and Operations
Viju Menon was named group president, global quality and operations in April 2018.
-
Acetabular System
Acetabular System
Have confidence in a powerful combination–a system that builds on the legacy that has defined our Trident brand for more than two decades, paired with the latest additive manufactured Tritanium In-Growth Technology or PureFix HA.
Trident II is created for case-to-case confidence that comes from building on a system backed by years of successful clinical data1-5 and reliable features such as the notable Trident Innerchange Locking Mechanism, Modular Dual Mobility (MDM), and our X3 precisely engineered polyethylene. -
Triathlon® Cementless – Fixation you can trust.
Fixation you can trust.
Triathlon Cementless combines the kinematics of Triathlon with the latest highly porous biologic fixation technology. With over one million cementless knees implanted to date1 and impressive survivorship data2-4 since it was introduced in 2013, Triathlon Cementless has become a trusted solution for surgeons across the globe.
-
Spine
We deliver the total spine experience, empowering our customers to keep the world moving. Together, we do more to challenge the standard, respond to clinical needs and pioneer new possibilities.
Our core technologies include biologic solutions, spinal implants, and surgical instruments for use in spine procedures. Our enabling technologies include digital health, guidance, and imaging for both cranial and spine procedures.
-
Global quality
At Stryker, quality is first in everything we do. Read our Quality Policy and learn more about how we build quality into our culture, our processes and our products.
-
Knee
Implants
We offer knee replacement implants for partial and total knee arthroplasty for primary and revision procedures. Our implants feature our flagship cemented and cementless TKA solution, the Triathlon Knee System.
-
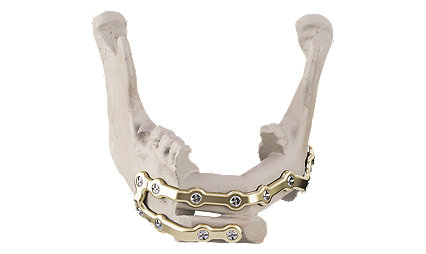
Facial iD - Mandible Reconstruction
A patient specific solution
Rigid internal fixation for primary and secondary mandible reconstruction.
-
Monterey AL Interbody System
Fixated on science
With a design to enhance the ability to create indirect decompression, a Tritanium structure to facilitate bone in-growth [1], and an array of footprints, heights and lordotic options — our Monterey AL Interbody System aims to deliver on all fronts. Plus, our instrumentation is designed to support your surgical style whether it’s freehand, partially guided or fully guided. Here’s a look at the careful crafting we do to help meet your surgical needs and personal preferences.
-
Environmental programs
Climate and the environment
Environmental programs, processes and initiatives to drive sustainability
-
Triathlon® Hinge
Introducing the newest addition to the Triathlon Knee Revision System
Triathlon® Hinge, the newest addition to the Triathlon Revision Knee System, has been designed to be a single platform for surgeries in patients with significant bone loss and/or ligament deficiencies.
-
Prime Series | Electric hospital stretchers
Stryker's hospital stretchers
Our Prime Series stretchers are designed to reduce physical strain on clinicians, enhance comfort during patient transport and drive hospital efficiency. Transporting heavier patients in a hospital gurney is now easier with Stryker's advanced mobility, while electric functionality gives patients the independence to adjust themselves without calling for caregiver assistance. Fully equipped and highly configurable, our Prime Series stretchers allow you to create a solution that meets your specific needs.
-
Champion SlingShot
Single Portal Suture Passing System
The Champion Slingshot has been optimized for use in the shoulder and allows you to both pass and retrieve suture using a single portal. Its ergonomic thumb-slide design helps to eliminate additional procedural steps and potentially reduce operating time.
-
Pivot Guardian total hip system
State-of-the-art surgical table accessories spanning six hip procedures
The Pivot Guardian total hip system includes the Pivot Guardian post-free distraction system for hip arthroscopy and now expands your options to five additional hip procedures with additional state-of-the-art table accessories.
-
Operon D830
Surgical table
The Operon D830 is designed to deliver on the increasing demand for flexibility in your operating room.
-
Emergency Care Training and Education
Discover on-demand webinars, online and in-person training and additional resources created specifically for EMS, first responders, hospital clinicians, the community and more.
-
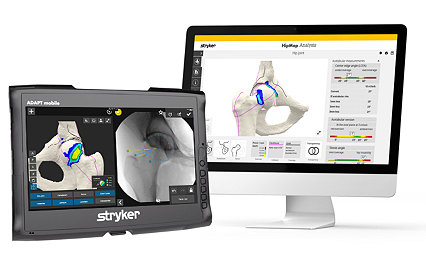
HipMap and HipCheck
Patient-specific surgical planning
3D preoperative planning assists with clinical decision making, and real-time intra-operative feedback helps measure and resect cam lesions to a desired alpha angle.
-
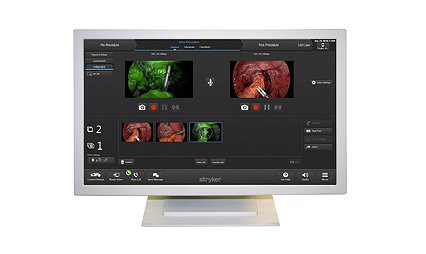
iSuite live video
Live video conferencing platform powered by ConnectSuite
iSuite live video connects clinicians within the operating room to other ORs, pathology for lab verification, surgeons for intraoperative consultations, and to auditoriums for education.
-
Teletom
Equipment management system
The Teletom equipment management system provides ceiling-mounted solutions to manage your surgical equipment, reducing reliance on carts and enabling you to streamline the workflow of your operating suites.
-
SpinePlex bone cement
SpinePlex bone cement is a derivative of Simplex P bone cement and has an average working time of 10-12 minutes. It contains the same Polymethylmetacrylate (PMMA) and is manufactured using the same proprietary process.
-
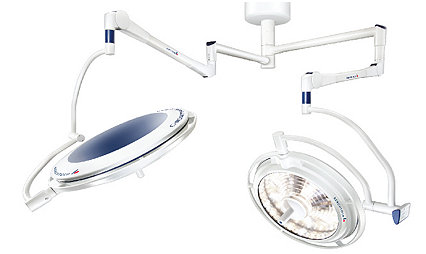
Berchtold LED F Generation
Surgical light
Illuminate the surgical field in your operating suite with our Berchtold LED F Generation surgical light. Delivering maximum brightness and offering multiple configuration options, you can customize your lighting experience.
-
Unique Device Identification (UDI)
The U.S. Food and Drug Administration (FDA) created unique device identification, often abbreviated UDI, a rule that requires medical device manufacturers to update their products with a unique device identifier that includes both device and production identifiers (such as expiration date and lot or serial number).
-
X3
Precisely engineered polyethylene
Our X3 polyethylene supports our total hip, knee and shoulder arthroplasty solutions. X3’s patented manufacturing process of sequential crosslinking uses three separate gamma irradiation doses with an annealing step after each.
-
GraftJacket NOW
Acellular Dermal Matrix
GraftJacket NOW is a ready-to-use, acellular human dermal matrix that provides strength and support with demonstrated vessel infiltration – a key indicator for graft incorporation.1 With its strong and flexible design and beneficial biological characteristics, it serves as a strong addition to strengthen primary repairs short and long term, eliminating the morbidity associated with harvesting allograft.
-
News archive
2014
-
Inbone
Total ankle system
The Inbone total ankle system is a modular prosthesis installed via the first and only intramedullary alignment system.

















?$preset_426_254$)