Customer Updates: Stryker Network Disruption > Read Now
Training and Education You specialise in your patients, we specialise in you.

Stryker delivers high quality personalised education programs designed to advance surgical techniques.
Healthcare professionals can count on us to help contribute to their success and drive value through training focused on quality clinical and technical application.Whether it’s at a local, national or European level, we are proud to provide a specialised educational experience.
Your opinion counts. We collect feedback after every session and the advice you give helps to make our programs even better.
A meaningful experience
________________
Make the most of your time. We strive to provide you with useful courses that optimise your learning.
An effective experience
________________
Stay in the know. Take what you learn here today, and apply it tomorrow.
A personalised experience
________________
We'll keep up. As you progress, so will our offerings.
Invested in you in
2019
Events
152
HPCs trained
2740
Stryker’s Medical Education focuses on the training and education of healthcare professionals on the safe and effective use of our products. We provide personalised support at two types of educational events:
Stryker Educational Events:
Educational events which are organised and run by Stryker, where we are responsible for the faculty, agenda and delegates.
Third Party Organised Educational Events:
Educational events where a third party (usually the course director or a university) is responsible for the faculty, agenda and delegates. Stryker can provide support in the form of medical education equipment and grants.*
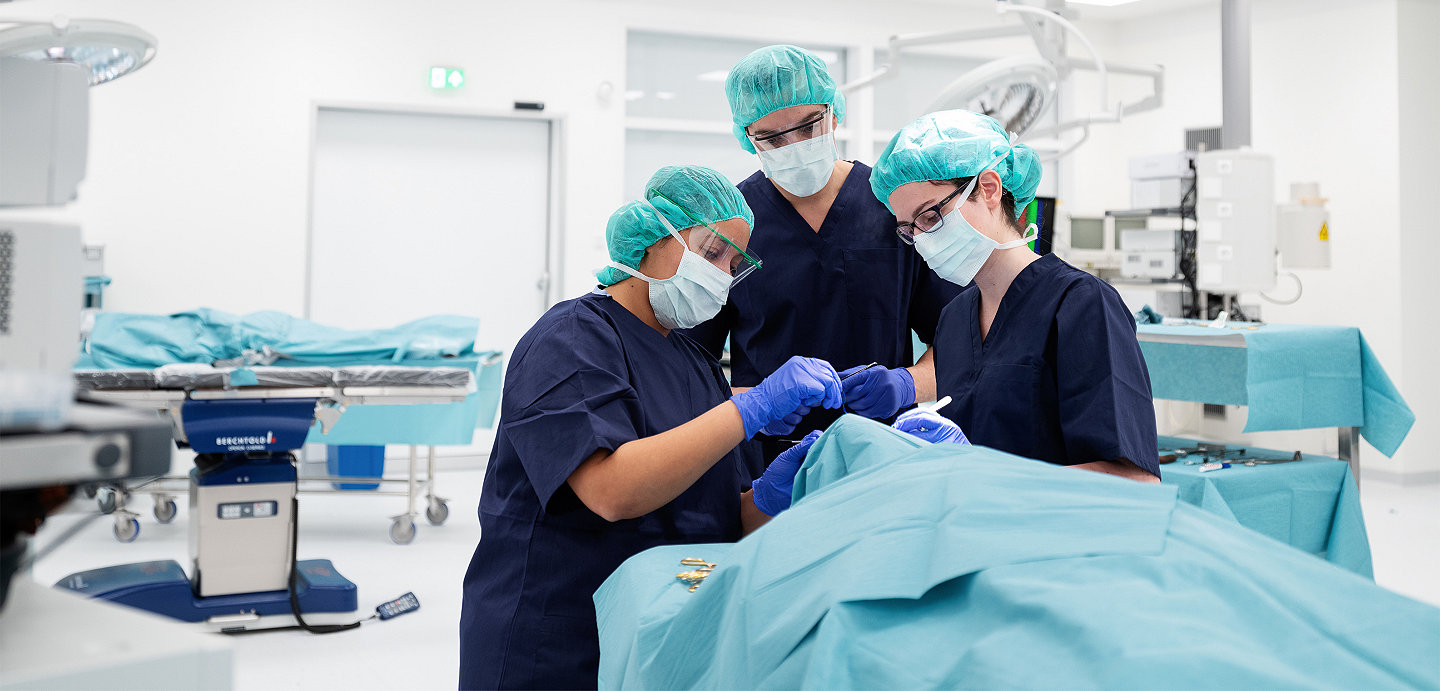
Your need for tiered levels of training and education is met by our extensive Stryker course offering ranging from fundamental resident training to advanced surgical techniques.
Essential
_____________________________
Education and training for healthcare professionals in the first years of their specialisation.
The opportunity to participate in local courses and events that showcase the safe and effective use of Stryker products.
Advanced
_____________________________
For healthcare professionals in their final years as a trainee and first five years in pratice.
Advanced level educational opportunities that reflect their increased knowledge and experience.
Expert
_____________________________
For senior level healthcare professionals.
The ability to enhance their knowledge with a view to positively impact the future of surgery.
Surgical training lab
Cadaveric labs focused on procedure and product education.
Clinical workshops
Simulations and training with models or animal bones.
Speaker series
Didactic based education.
Broadcast
Live streaming educational events including surgery and symposiums.
Surgeon Observation
In theater visitation program.
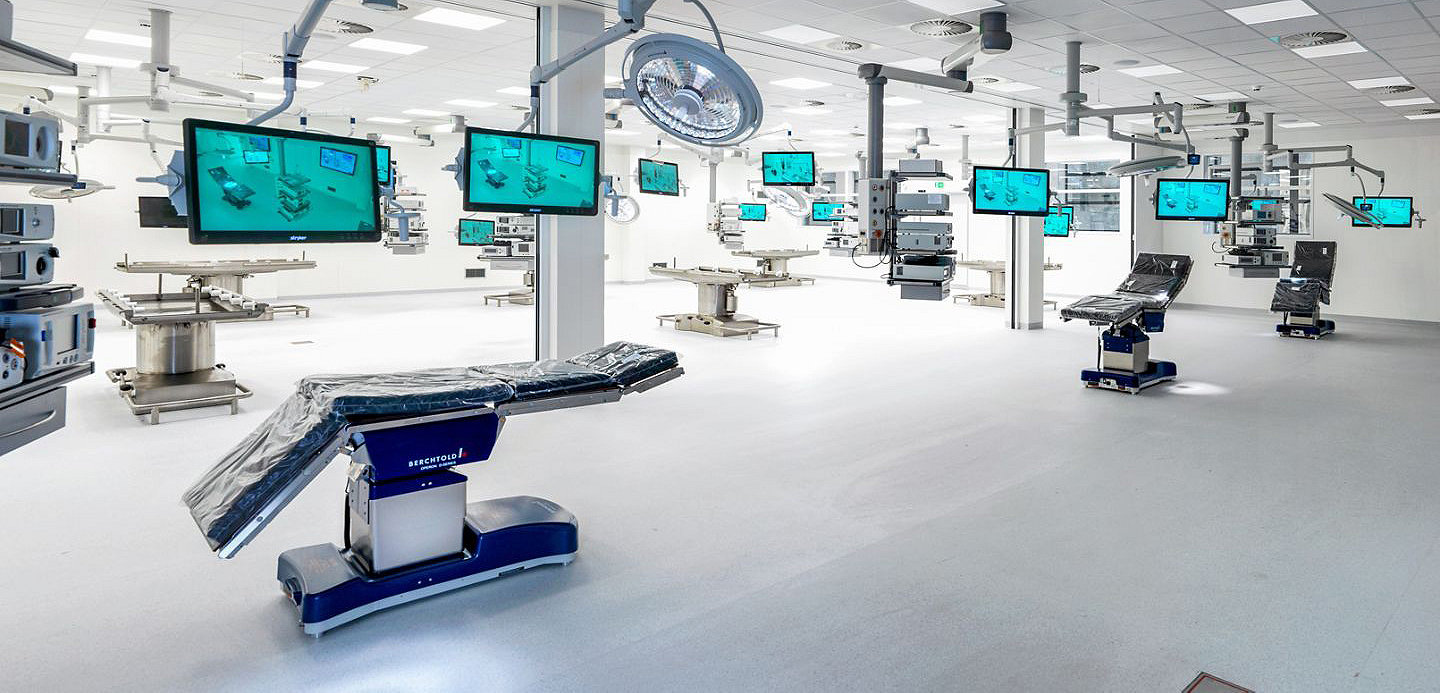
On 4th February 2019, the Amsterdam Skills Centre was opened in the Netherlands. Stryker is a proud partner together with the Amsterdam University Medical Center (AMC) and Vrije University (VU).
The Amsterdam Skills Centre provides a state of the art operating theater, with 12 operating tables providing training of complex procedures on preserved human tissue. A digital learning environment is supported in the 3700m2 building, which has conference rooms for up to 200 people.

Stryker is a member of MedTech Europe, which is the European Industry association representing the medical technology industries, from diagnosis to cure.
As a member of MedTech Europe, Stryker is committed to comply with the Med Tech Europe Code of Ethical business Practices while partnering on educational opportunities.
For further information, please visit the MedTech Europe website: www.medtecheurope.org
EANS research grant
The Stryker EANS research grant aims to promote clinical research into diseases of the brain and spine in Europe by providing seed grants to jump-start projects in the Neurosurgical field.
While major advances have been made in the treatment of brain injuries, brain tumours, stroke, epilepsy, hydrocephalus and disorders of the spine, there remain many unanswered questions and major opportunities to improve treatment and outcome for patients.
Stryker has committed to partnering with EANS to address these opportunities by supporting a grand of €10.000. The grant is intended to enable the acquisition of the pilot data necessary to apply for the substantial funding required to complete such projects from large grant funding bodies.
Interested parties should download and submit an application form, which can be found at the EANS website:
https://www.eans.org/page/EANSResearchFund2020
Deadline December each year

*subject to compliance approval and MedTech Europe guidelines.
2019-22548

.pdf.thumb.319.319.png)