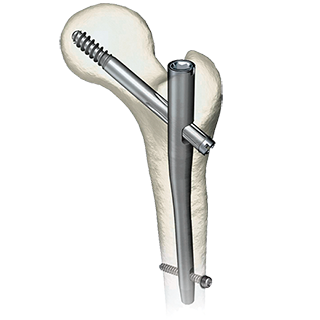
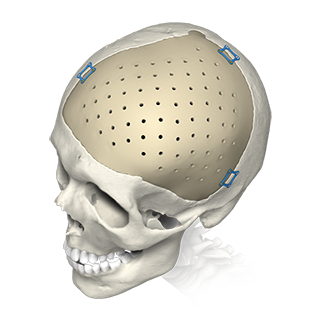
Customized Mandible Recon Plate KitIntended to be used for rigid internal fixation of primary and secondary mandibular reconstructions.
Focused on
better

Above:Dustin “Doc” Kirby and Brandi smiling big.
Iraq veteran
faces his new life
Almost 10 years after he was shot through the mouth, Dustin “Doc” Kirby, a veteran of the Iraq War, was having his face operated on — again — in 2016.
David L. Hirsch, D.D.S., M.D., F.A.C.S. Chief, Oral-Maxillofacial Surgery, Department of Otolaryngology — Head & Neck Surgery, Lenox Hill Hospital

Just before the procedure, the surgical team, led by David L. Hirsch, D.D.S., M.D., F.A.C.S., removed the metal hardware — rows of titanium screws and plates — that had held Doc’s previously shattered bones in place.
With the surgery, the goal was to change how Doc looked, ate, talked and smiled, essentially changing his life from a traumatized, disfigured man to the spirited, healthy father of four he wanted to be again. The team’s objective was to realign Doc’s profile and bite using new screws and a customized, patient-specific titanium plate.
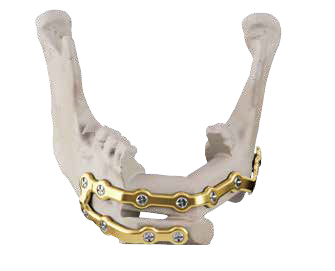
Customized Mandible Recon Plate KitIntended to be used for rigid internal fixation of primary and secondary mandibular reconstructions.
On Christmas Day 10 years ago, when Doc was 22, a single bullet entered through his left cheek and exited his right, ripping out part of his tongue and seven teeth, shattering the right side of his jaw and cracking part of his lower skull near the roof of his mouth.
The first medical team on the case saved his life. In subsequent years, which included 30 surgeries, doctors focused on the slow process of reconstructing his face. Doc began speech therapy, so he could relearn to talk, and psychological counseling to deal with post-traumatic stress disorder and his adjustment as a wounded warrior. He continued to struggle with chronic pain, migraines, insomnia and a rebuilt jaw that did not align with his teeth.
“We were at a point where there was not much more they could do,” Doc says, “but I wanted my face back.”
Our customized mandible reconstruction technologyEventually, through a veterans program, Doc found Dr. Hirsch, who was one of the pioneers of virtual surgical planning (VSP) with 3D Systems. This technology is used to design and create three-dimensional models, along with 3D printed cutting and positioning guides of the patient’s current and reconstructed anatomy. The technology, provided by 3D Systems, was not available when Doc was originally treated.
“It’s such an
amazing, fantastic
change. It’s like
I’m brand new.”
Our Customized Mandible Reconstruction Plates are designed by using proprietary software and can be used in conjunction with 3D Systems’ VSP technology that incorporates data from a CT scan. The patient-specific plates are designed based on inputs from a surgeon to treat each patient’s unique condition.
“With this web-based technology, we have the ability to visualize anatomical landmarks to make sure the surgical plan is as accurate as humanly possible,” explains Dr. Hirsch. “Cutting and positioning guides help to transfer the surgical plan into the operating room.”
Dr. Hirsch also used our Colorado MicroDissection Needle, an ultra-sharp tool designed for simultaneous cutting and coagulation, which reduces local blood loss and promotes a clearer surgical field.
Weeks after the surgery, Dr. Hirsch says, “I don’t think there will be any restrictions on what he can do. He’s happy and pain free, and I’m thrilled with his progress.”
“I couldn’t imagine a life without pain, but here I am feeling great,” Doc says with a big smile. “I used to spend a lot of time looking in a mirror and not seeing me,” he adds. “It’s such an amazing, fantastic change. It’s like I’m brand new.”
Note: Dr. Hirsch is a paid consultant of Stryker. His statements represent his own opinions based on personal experience and are not necessarily those of Stryker. Individual results will vary.
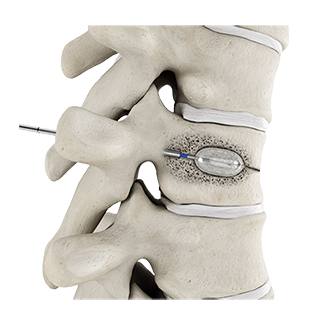
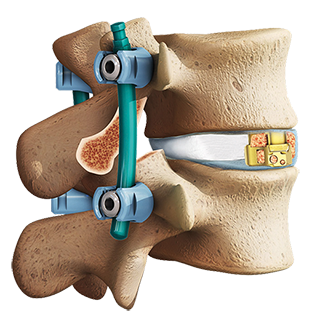
Tritanium PL Cage

Additive
manufacturing
AMagine is our proprietary approach to implant creation using additive manufacturing, also known as 3D printing. AMagine technology is the result of our 10+ years of experience, expertise and excellence in the manufacturing of surgical implants.
Thanks to AMagine, today we are able to tackle previously insurmountable design complexities, address unmet surgeon needs and make and sell more metal implants developed using additive manufacturing than any other company in the world. Other benefits of additive manufacturing include the ability to produce minimal material waste and the potential to offer unrivaled product development speed and thanks to the fact that each implant is generated from a single master computer model, the ability to create a metal porous matrix for a given model and size of a particular product that is virtually identical to that which was originally tested and validated. Some examples of new implants manufactured using our AMagine approach include the Triathlon Tritanium Cementless Baseplate and the Tritanium Posterior Lumbar (PL) Cage.
2016 acquisitions
focus on prevention
In 2016, we completed the acquisition of two leading companies — Physio-Control and Sage Products — that are focused on preventing medical complications and improving patient outcomes.
With the acquisition of Physio-Control — which develops, manufactures and markets monitors/defibrillators, automated external defibrillators (AEDs) and CPR-assist devices along with data management and support services — we are strengthening our scope and capabilities in the public access, emergency medical services (EMS) and acute care markets. Physio-Control has a long history of innovative products that are specifically designed to improve patient outcomes and increase cardiac arrest survival rates.
In addition, Physio-Control has achieved a global leadership position with a strong brand known for durable, high-quality products that predict or intervene in life-threatening emergencies, which will expand and strengthen our global presence.
Sage Products’ offerings include solutions for oral care, skin preparation and protection, patient cleaning and hygiene, turning and positioning devices and heel care boots. Many of these disposable products are targeted at helping to reduce “never events,” primarily hospital-acquired conditions in the intensive care and MedSurg unit settings. Sage Products’ success is reflected in its 12-year run of double-digit sales growth.
In 2017, we will promote Sage Products’ market-leading Prevalon franchise, which focuses on helping to address pressure injuries and patient handling injuries, as well as comprehensive oral hygiene systems to complement our existing acute care business.

Physio-Control's emergency care medical products, LIFEPAK 15 Monitor/Defibrillator and LUCAS 3 Chest Compression System.
Physio-Control's emergency care medical products, LIFEPAK 15 Monitor/Defibrillator and LUCAS 3 Chest Compression System.

Above:Sage Products offers prevention solutions that help reduce the risk of “never events.”
Mako Robotic-Arm
Assisted Surgery
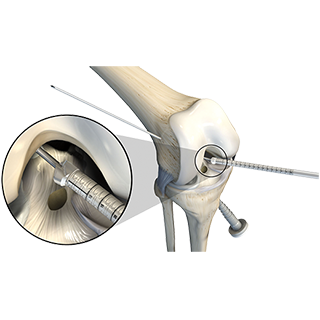
Mako Robotic-Arm Assisted Surgery enables surgeons to treat knee and hip conditions with greater predictability1,2,3 through more accurate component placement and reproducibility in both partial knee and total hip replacement.

Above:Mako Total Knee
In the United States robotic-arm assisted surgery is associated with lower all-cause readmission rates for unicompartmental knee arthroplasty procedures within 30 days (over 36% lower) and 90 days (over 16% lower). These reduced readmission rates translate to 40% lower readmission costs at 30 days and 66% lower readmission costs at 90 days.4
The Mako System offers Total Hip, Partial Knee, and now Total Knee applications. The demand for primary knee replacement procedures alone in the United States is expected to increase 673% by 2030.5
We celebrated the full commercial launch of the much-anticipated Mako Total Knee application in March 2017, which combines Mako technology with the clinically proven Triathlon Implant System to offer a truly innovative solution for total knee replacement
- Blyth M, Jones B, MacLean A, Anthony I, Rowe P. Accuracy of UKA implant positioning and early clinical outcomes in a RCT comparing robotic assisted and manual surgery. In: Proceedings 13th Annual Computer Assisted Orthopedic Surgery Meeting; June 12-15, 2013; Orlando, FL, USA. (Blyth/Jones 2013b).
- Coon T, Roche M, Buechel F, et al. Short to mid term survivorship of robotic arm assisted UKA: a multicenter Study. In: Proceedings Annual Pan Pacific Orthopaedic Congress; July 16-19, 2014; Kona, HI.
- Domb BG, Bitar YFE, Sadik AY, Stake CE, Botser IB. Comparison of robotic-assisted and conventional acetabular cup placement in THA: a matched-pair controlled study. Clin Orthop Relat Res. 2013; 472(1):329-336.
- Baker Tilly, LLP. Mako Robotic-Arm Assisted System: A Clinical and Economic Analysis for Health Plans and Providers. 2016
- Kurtz AAOS. Total knee and hip replacement projections 2030. www.prnewswire.com/news-releases/total-knee-and-hip-replacement-surgery-projections-show-meteoric-rise-by-2030-55519727.html. Accessed July 4, 2016.
lower readmission costs at 90 days
Ryan McCarthy Managing Director, Medical, Australia and New Zealand

Ryan McCarthy honored for
transformational leadership
Gallup Great Workplace Award winners represent the epitome of an engaged and high-performing workplace, and we are proud to be a nine-time winner.
Gallup also gives organizations the opportunity to nominate someone for the Manager of the Year award, with a panel of workplace experts selecting a winner from the pool of outstanding nominees. For 2016, we nominated Ryan McCarthy, Managing Director, Medical, Australia and New Zealand, who was among the top 10 finalists for the award.
The nominees are consistently in the 90th percentile or above in overall engagement, have shown high levels of performance, attain high retention of employees and achieve consistent engagement scores over time. Ryan increased engagement across all the teams he has led, which puts him in the 97th percentile. Under Ryan’s leadership, his team dramatically increased the number of government hospital contracts over the last several years.
“These results are the natural outcome of a leader who gives his people the freedom to do what they do best every day, while supporting and inspiring them to constantly raise the bar,” says Maurice Ben-Mayor, President, South Pacific. “Ryan’s passion and enthusiasm are contagious, and his team takes that mindset into every customer interaction, always committed to finding ways to make other teams and the products and services we provide to our customers better.”
“Ryan’s passion
and enthusiasm
are contagious
and his team takes
that mindset into
every customer
interaction.”
Recognizing
R&D excellence
In 2015, we introduced the R&D Fellows Award to honor five individuals for their significant accomplishments in research. Last year, we inducted two more Fellows — John Daniel, Vice President, R&D, Neurovascular and Mike Steffler, Vice President, Global R&D and Innovation, Medical.

John Daniel (left) & Mike Steffler (right)

John Daniel (left) & Mike Steffler (right)
John Daniel
John has worked in the field of less invasive interventional medicine for more than 25 years. He has made significant contributions to the Neurovascular business, having launched over 20 new products globally in the last five years, resulting in double-digit growth in 16 of the last 20 quarters for the division. One of his many contributions was launching the successful Target Detachable Coil. John has been awarded 22 patents, with 10 applications pending.
Mike Steffler
Mike has more than 37 years of technical design and leadership experience in such market segments as endoscopic and orthopaedic surgery, medical and surgical, ICU clinical environments and pre-hospital patient care. He is currently co-leading Customer Centered Design methodology implementation across the businesses in Kalamazoo, Michigan. In 2011, he headed the Corporate Innovation Initiative, which led to the introduction of such new approaches as Ten Types of Innovation.
Collaborative education,
specialized learning
Our new learning center, located in Salt Lake City, Utah, will be collocated with our brand-new neuro manufacturing and distribution facility located just minutes from downtown and the international airport.

The 7,800-square-foot, state-of-the-art facility was developed with the latest integration technology complete with a fully functional iSuite, integrated bio-skills lab, interactive classroom and video conferencing rooms. This center will offer an unmatched setting where our customers can interact with our sales, marketing, product innovation and medical education teams in a facility optimized for learning in a highly specialized operating environment. The center is scheduled to open in the first quarter of 2017.